No 28/33 - 2022
Update on the monkeypox outbreak in Europe
Update on the monkeypox outbreak in Europe
Since the latest issue of EPI-NEWS on monkeypox (25/2022), the Danish Health Authority (DHA) has amended its guidelines on handling of monkeypox, primarily changing the case definition and recommendations on infectious hygiene precautions and vaccinations. Furthermore, Statens Serum Institut (SSI) has published a new risk assessment. Therefore, this EPI-NEWS was extended to also include these issues.
Background
To this day, Denmark has recorded a total of 155 cases of monkeypox. In the early phases of the outbreak in Europe, persons who had monkeypox detected in Denmark had become infected during travels to other European countries. Since July, transmission has mainly occurred in Denmark. All of those infected are men, see Figure 1. A cluster of cases is observed among men who have sex with men (MSM) and who have several sexual partners. Even so, the risk of becoming infected is not linked to gender or sexual orientation, but to a contact pattern including frequent close physical contact within a group where the infection circulates. Therefore, it is important to stress that anyone who comes into close physical contact with a person who has become infected is at risk of infection.
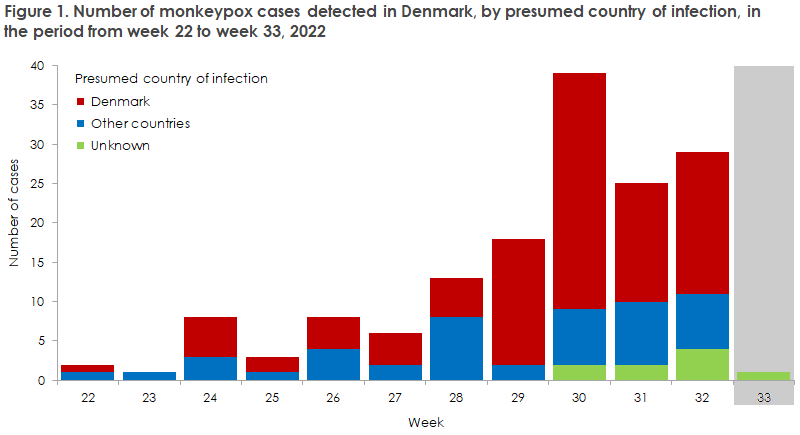
In the short term, the SSI assesses that in the absence of any preventive measures, there is a very high likelihood that monkeypox will spread further and gain a foothold among MSM with many sexual contacts. The risk is also assessed to be high for other groups with many sexual contacts, but low for healthcare workers and very low for pregnant women, children, immunosuppressed people and the remaining population. Vaccination of MSM with risk behaviour will reduce the risk that the infection gains a foothold among MSM and will probably also reduce the risk of spreading to other population groups.
As approx. 50% of the persons who have been tested for monkeypox have tested positive and as infected persons may have mild symptoms, we expect that a number of unrecognised cases exist. Healthcare workers are therefore encouraged to pay extra attention to relevant symptoms and to test more than previously. Persons who present with symptoms that may raise a suspicion of monkeypox should be swabbed for monkeypox.
On primary suspicion of genital herpes, syphilis or gonorrhoea in a person who has engaged in sexual risk behaviour, testing for monkeypox as a differential diagnosis should be considered.
Guidelines on the handling of monkeypox in Denmark
The Danish Health Authority has published an amended guideline describing the handling of monkeypox in Denmark. The guideline includes a thorough description of the condition, including the
notification procedure, case definition, hygiene precautions, contact tracing, treatment and vaccination.
Case definition for a suspected case
In Denmark, monkeypox is suspected in a person in case of:Unexplained rash on the skin or in mucous membranes OR one or more other symptoms that may indicate monkeypox*AND as a minimum one of the following:
- Epidemiological connection to a confirmed or suspected case of monkeypox within 21 days leading up to the person’s symptom onset OR
- Travel activity to an area where monkeypox occurs endemically** within 21 days before symptom onset
OR
- If the patient, regardless of sexual orientation, has had several or anonymous sexual partners within 21 days before symptom onset
OR
- If the patient is a man who has sex with men
*Influenza-like symptoms, including a fever > 38.5 °C, headache, back pain, fatigue, lymphadenopathy (locally or generally) and proctitis
**Central and West Africa
Symptoms
Monkeypox typically presents as fever, myalgia, headache and swollen lymph nodes, followed by a rash with blisters (vesicles). Read more in the Disease Encyclopaedia (Sygdomsleksikon).
The current outbreak has seen a predominance of cases in which the rash presents around the genitals and on the buttocks, and it may be limited to these areas. Some patients develop a very painful rash and/or inflammation in and around the rectum. The rash may be limited to very few or even a single element and may in some cases be absent. Similarly, general symptoms may be absent. Pain, and in some cases haemorrhage from the rectum due to local inflammation, has been described as the only observed symptom. In persons who develop a rash in and around the rectum, the pain may be so pronounced that analgesic treatment and in some cases admission to hospital may be needed.
Differential diagnostic considerations should, among others, include chickenpox, syphilis and herpes, the symptoms of which may be mistaken for monkeypox. Additionally, symptoms from the rectum may be mistaken for gonorrhoea.
Infectious hygiene precautions at hospitals, in general practice and in the emergency call service - suspected case
General preautions
- The patient is placed away from other persons, if possible in a one-bed room, and should only see staff who need to come into contact with the patient.
- A standard surgical mask is handed out. The mask is placed over the face by the patient him- or herself.
- If possible, the patient’s medical history and general condition are assessed from a distance. You may ask the patient to take his or her temperature using an oral thermometer, which is subsequently discarded.
Personal protection gear
- If close or direct contact to the patient or the patient’s surroundings is needed, the staff should carry personal protection gear in the form of gloves; long-sleeved, liquid-repellent single-use gown with cuffs; a surgical mask and eye protection (full-coverage face mask/protective goggles).
- In case of increased risk of transmission by aerosol (from coughing - including during throat swabbing, prolonged handling of patients, changing of bed linen, etc.), an FFP3 mask should be used rather than a surgical mask, based on a precautionary principle.
Cleaning - Cleaning is done as usual.
Sampling
Swabbing may be done at an infectious disease department, a dermatology department/clinic for sexually transmissible diseases, at a general practitioner or in other hospital departments/wards, as relevant. You may want to confer with an infectious disease department.
If an indication for monkeypox testing is found, one or more of the following is taken:
- Swabs from skin lesions, the rectum and the throat.
- Vesicle liquid (50-100 micro litres).
Biopsy: In case of relevant exposure, a test should also be offered for sexually transmitted infections, along with relevant differential diagnostics.
- Vesicle: From a skin vesicle, 50-100 micro litres of vesicle liquid is sucked up using a small syringe/needle (e.g. 0.5 mL insulin syringe) and transferred to a dry vial.
- Swabbing/biopsy/scraping: Swabs are collected with a swab. The swab/biopsy/scrapings are submitted in a dry vial. When vesicles are swabbed, we recommend that a few vesicles are swabbed with the same swab to ensure that a sufficient amount of virologic material is collected for PCR diagnostics.
- Swabbing from various anatomical localisations is done with separate swabs and submitted in separate test vials, ensuring that each vial states the localisation where the sample was taken.
Laboratory diagnostics
Samples may currently be analysed at the Department of Virology, Statens Serum Institut; the Department of Clinical Microbiology (DCM) at Rigshospitalet, Aarhus University Hospital, Skejby; Alborg University Hospital South and at Odense University Hospital (OUH).
If a DCM cannot perform the analysis, an agreement may be made with another DCM close by to do the analysis, or the sample may be submitted to the SSI. If the sample is submitted to the SSI, it must be packed in a primary container (test vial, lance tube) and in a secondary container (transport vial). The primary and the secondary container must be separated by an absorbent material capable of absorbing all of the sample liquid. The sample is packed in suitable external packing material to ensure the sample against external blows. The sample envelope is marked “Note: Monkeypox” (In Danish language: “Obs. Monkeypox”).
All positive samples must be sent from the analysing DCM to the SSI for genome sequencing, etc.
Duty to notify
Any confirmed case of monkeypox must be notified immediately (on the same day) by the treating department/physician. Notification is by phone to the Danish Patient Safety Authority (DPSA). Shortly thereafter, the case must be notified in writing via the Danish Health Data Authority's Electronic Notification System (SEI2) to Statens Serum Institut (SSI), Department of Infectious Disease Epidemiology and Prevention. These notifications are used for infection tracing, for assessment of the need for vaccination of close contacts and for epidemic monitoring purposes, including reporting to the European Centre for Disease prevention and Control (ECDC), which is done twice weekly. Therefore, it is important that notification to the DPSA and the SSI alike is completed upon confirming a positive test result.
The DPSA is responsible for ensuring contact tracing of close contacts to persons with confirmed monkeypox infection. The DPSA contacts the relevant department of infectious medicine which then assesses if post-exposure prophylaxis (PEP) should be offered in the form of vaccination of close contacts.
Vaccination
A live, attenuated vaccine is available, which may be used for protection against monkeypox (Imvanex/Jynneos). The vaccine is approved for use in persons aged 18 years or older and may be used either before the exposure (prophylactically) or after a person has been exposed to infection (post-exposure prophylaxis - PEP). Following medical assessment, the vaccine may be used in special groups, including children, provided the risk of serious disease outweighs the risk associated with the vaccination.
On 25 July 2022, the European Medicines Agency (EMA) authorised the Imvanex vaccine for protection against monkeypox. The vaccine has been authorised since 2019 for the same use by the US medicines agency (FDA), also see the SSI Vaccine Encyclopaedia (Vaccineleksikon). As the vaccine is also marketed under the name of Jynneos, it is important to enter the vaccine in the Danish Vaccination Register (DVR) using the correct product name.
The Danish Health Authority has assessed that to prevent serious disease, vaccination may be offered to close contacts with a moderate to high risk of becoming infected. The vaccination offer is given only to close contacts following specific and individual assessment by an infectious medicine specialist. The medical assessment takes into account the risk of infection and the risk of developing serious disease.
On 9 August 2022, the Danish Health Authority extended the vaccination offer.
Vaccination may be offered to the following groups of persons with risk behaviour that increases the risk of infection:
- MSM who are taking PrEP (preventive medication) against HIV infection.
- MDM who are not taking PrEP against HIV, but who meet the criteria in place to be given such medication. This may include persons who belong to the above target group of MSM, but who do not tolerate PrEP against HIV, have opted out of receiving PrEP against HIV or are receiving HIV treatment.
When assessing the target group of persons who are at high risk of infection, the following criteria should be considered. The criteria are equivalent to those that apply to persons who receive PrEP against HIV:
- MSM who have had unprotected anal intercourse with a minimum of two male partners within the past 12 weeks (regular known HIV-negative partner is not included).
- MSM who have had syphilis, chlamydia or gonorrhoea within the past 24 weeks.
Women or transgender persons who form part of the same network as MSM who are at a high risk and who have a similar contact pattern may also benefit from preventive vaccination. If a medical assessment based on the above-mentioned criteria determines that other persons may be considered to fall within the target group of persons who are at an increased risk of infection, they may also be offered preventive vaccination.
The Danish Health Authority has worked jointly with the Danish regions to plan how the practical implementation of the vaccination offer will be. The regions are responsible for giving the vaccinations and for providing information for persons who are at a special risk about where and how they may contact their region to be vaccinated. Vaccination of persons at a special risk was initiated on 12 August 2022.
Persons who are taking preventive PrEP treatment against HIV will receive a letter inviting them to be vaccinated. The letter will also provide specific information about the vaccination.
Persons who are not taking PrEP treatment, but who meet the criteria for being offered vaccination may find information at the website of their region explaining where they may go to be referred for vaccination against monkeypox. In the Capital Region of Denmark, these persons should see their general practitioner, who may refer them to a department of infectious diseases.
For further information, please see the guidelines of the Danish Health Authority on handling of monkeypox in Denmark.
Monkeypox virus and human poxvirus
Monkeypox is caused by a rare virus found in some African animals. The virus can also be transmitted to humans. Monkeypox virus is related to human poxvirus, which was eradicated globally in 1980, and which was transmitted between humans exclusively and was not found in animals. Various African countries record monkeypox cases continually, but outbreaks outside of Africa are rare.
Monkeypox is less infectious than humanpox and it runs a less severe course. Monkeypox typically presents as fever, myalgia, headache and swollen lymph nodes, followed by a rash with blisters (vesicles). Read more in the Disease Encyclopaedia (Sygdomsleksikon).
The international situation
On 7 May 2022, Great Britain notified the WHO of a confirmed case in a person who had travelled from Great Britain to Nigeria and had subsequently returned to Great Britain. In Nigeria, the condition is widespread (endemic), and monkeypox was suspected. Therefore, the person was immediately admitted to hospital and isolated, and reverse transcriptase-polymerase chain reaction (RT-PCR) confirmed that the man had monkeypox (West African type).
On 16 August 2022, a total of 38,019 cases of monkeypox had been reported from a total of 93 countries where monkeypox does not occur endemically. Based on information from Our World In Data, an average 850 new confirmed cases are currently being recorded daily (as per 2 August 2022). This number is higher than the number recorded for the previous week, but slightly lower than the number recorded for the period from 30 July to 1 August. The countries with the highest number of cases are the US (12,688 cases), Spain (5,719 cases), Germany (3,186 cases), England (3,081 cases), Brazil (2,893 cases) and France (2,673 cases).
Overall, the European countries account for the majority of the cases with a total of 17,897 reported cases (according to the ECDC), corresponding to nearly two thirds of the cases reported globally. Cases reported in non-endemic countries primarily occur in men, who identify themselves as homosexuals, bisexuals or men who have sex with men (MSM), see the website of the English health authorities. Even so, cases have also been reported in women and children.
As per 10 August 2022, the ECDC and the WHO have reported that among the 17,286 cases in the European region, 172 are women (1%) and 30 are 0-17 years of age (0.17%). The majority of cases in Europe are adults aged 31-40 years of age. On 10 August 2022, the ECDC informed that MPX virus had then been detected in 48 healthcare workers, but none of these cases are believed to have been due to occupational exposure.
In a recent statement of figures from the TESSy monitoring scheme run by the ECDC, information was provided that the household was stated as the place of infection among 102 of 1,097 people infected with monkeypox (9%) for whom information had been provided about the place of transmission. A total of 78 of these patients also reported mode of infection: 83% reported sexual transmission and 15% non-sexual person-to-person transmission as the most likely mode of infection. Based on this, sexual transmission is, by far, the most prevalent mode of transmission, even within households. The same report found that 1 of 16 children aged 0-17 years with monkeypox had become infected by non-sexual contact within the household, whereas no information was provided regarding place of infection for the remaining 15 cases. The ECDC and the WHO do not report numbers for household transmission or transmission among sex workers.
-It is possible that the outbreak has been ongoing for a longer period of time, as monkeypox cases from Nigeria diagnosed in the US in November 2021 hold genetic sequences belonging to the same virus strain that is now being detected in Europe and in other countries.
The ECDC regularly publishes risk assessments.
On 23 July 2022, the WHO’s Director-General declared monkeypox a Public Health Emergency of International Concern (PHEIC), which means that monkeypox is “an extraordinary event that comprises public health risk in other countries due to the risk of spreading internationally, and which may require internationally coordinated action”, and that the event is either “serious, sudden, unusual or unexpected”. This does not necessarily mean that the condition is very serious. That a disease is declared a PHEIC means that the WHO encourages the individual countries to strengthen their efforts against monkeypox with respect to diagnostics, monitoring, treatment and prevention, and that the WHO may make a range of tools available to the countries.
The US also declared monkeypox a national health crisis on 4 August 2022.
Mortality
In an African context, a mortality rate of 3.6-4.6% was reported for the West African strain, which is the one currently observed in Europe, and up to 8.7% for the Central African strain. The observed mortality in Central and West Africa cannot be extrapolated directly to a general expected mortality in Europe as treatment opportunities in the African countries are poorer, the occurrence of immunodeficiency higher due to untreated HIV infection, malnutrition, etc, and because the diagnosed cases will represent the more serious cases. The cases observed during the current outbreak in Europe have generally been mild to moderate, and it is therefore assessed that unacknowledged cases will also have occurred.
In the latest update from the WHO (WHO Situation Update), five deaths in Africa were reported. On 29 July, the first deaths related to the monkeypox outbreak were reported from non-endemic countries; Spain (n = 2) and Brazil (n = 1), respectively. Subsequently, notifications of deaths have been recorded from Peru (n = 1) and India (n = 1). The current outbreak in non-endemic countries has been caused by the West African clade (subgroup of virus), which seems to cause less serious symptoms than the Congo Basin clade.
Only sporadic information has been provided about deaths, but with respect to the Brazilian death, authorities reported that the person was affected by lymph cancer and died with symptoms of sepsis. In contrast, the two Spanish cases were seemingly healthy when they became infected with monkeypox but died with symptoms of infection of the brain and the lining of the brain (meningoencephalitis), which was reported as a rare manifestation of monkeypox. No deaths caused by monkeypox have been recorded in Denmark.
Phylogeny
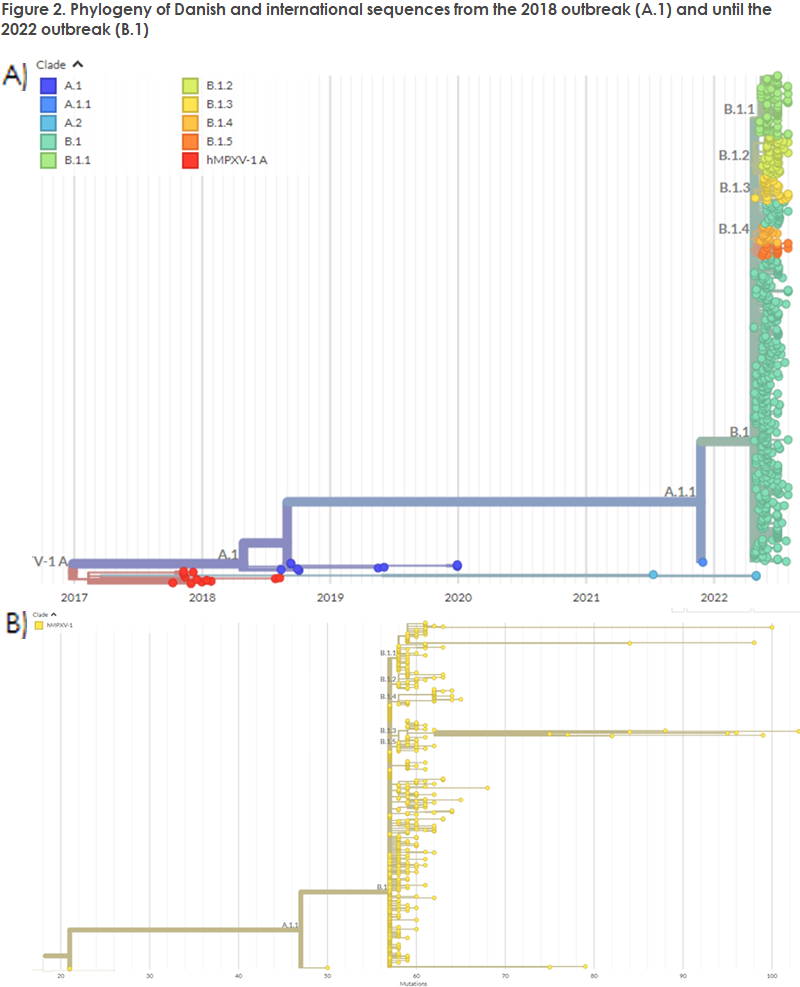
Until the current outbreak, the two clades of monkeypox virus were described using geographical names (the West African and the Congo Basin clade), but the use of geographical names to describe monkeypox variants has been changed as from the current 2022 outbreak and sequences will, from now on, be named clade 1 (formerly the Congo Basin) and clade 2 (formerly the West African clade). Furthermore, clade 2 has been divided into more detailed clades.
The new nomenclature describes hMPX outbreaks, where A.1 characterises the 2018 outbreak with the West African variant, and further branching to A.1.1, which comprises a single US case in 2021 and the new 2022 outbreak. The current 2022 outbreak was coined B.1, as the sequences form a shared cluster based on their genetics. Within the B.1 clade, an additional subdivision has been introduced comprising B.1.1-B.1.5.
It deserves mention that the subdivisions of clade B.1 do not describe new outbreaks or geographically delimited subvariants. Also, they do not describe variants that produce a changed disease course or have infectious potential. The Danish sequenced cases all belong to B.1, specifically to the sub-variants B.1.1 and B.1.3 (Figure 2, B). The placement in the phylogeny three of the US sequences from November 2021 may possibly indicate that monkeypox has been in circulation for a longer period than previously assumed, and it is now being proposed that it may have been in circulation since April 2016.
(A.K. Hvass, A. Koch, Department for Infectious Disease Epidemiology and Prevention and M. Bennedbæk, Virus and Specialised Microbiological Diagnostics)
17 August 2022